|
The calculation method reviewed above for determining formal charges on atoms is an essential starting point for a novice organic chemist, and works well when dealing with small structures. formal charge valence shell electrons (free atom) lone pair electrons 1 2 bonding electrons formal charge valence shell electrons (free atom) lone pair electrons 1 2 bonding electrons We can double-check formal charge calculations by determining the sum of the formal charges for the whole structure. All atoms in BrCl 3 have a formal charge of zero, and the sum of the formal charges totals zero, as it must in a neutral molecule. 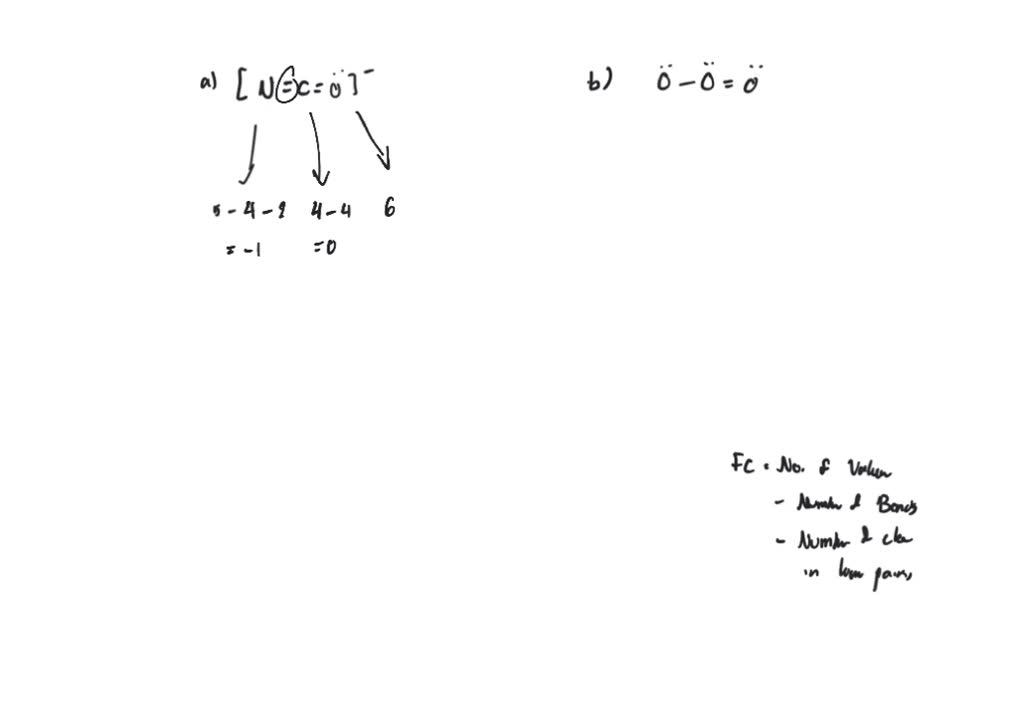
This gives the formal charge:Br: 7 7 0Cl: 7 7 0. Subtract this number from the number of valence electrons for the neutral atom. 
In this case, the sum of the formal charges is 0 1 0 0 0 = 1 , which is the same as the total charge of the ammonium polyatomic ion.ĭetermining the Charge of Atoms in Organic Structures Now each Cl atom has seven electrons and the Br atom has seven electrons. 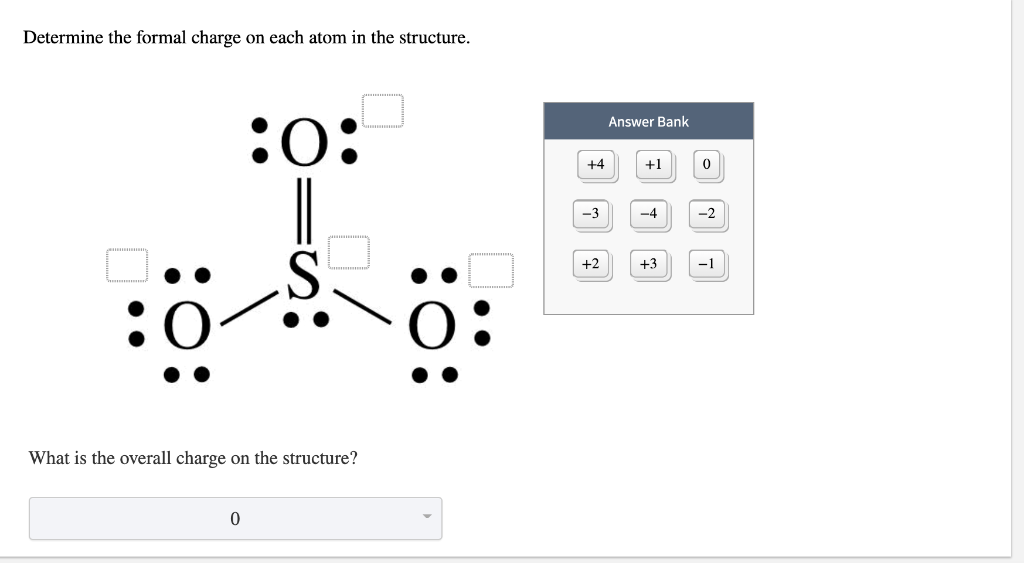
\) ion are thusĪdding together the formal charges on the atoms should give us the total charge on the molecule or ion. The formal charge can be calculated by excluding the number of electrons in the lone pairs and the number of bonds from the total number of valence electrons.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
